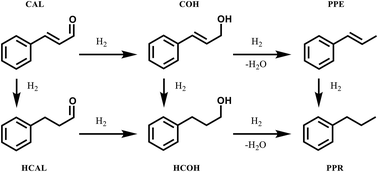
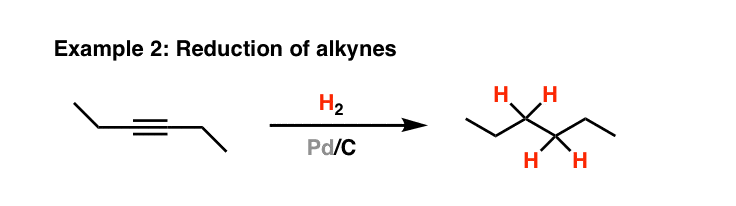
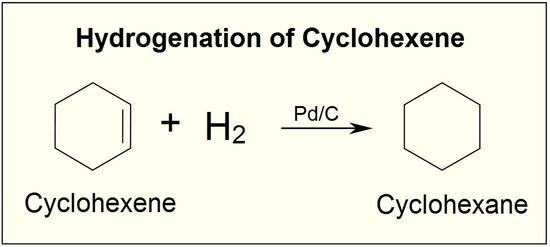
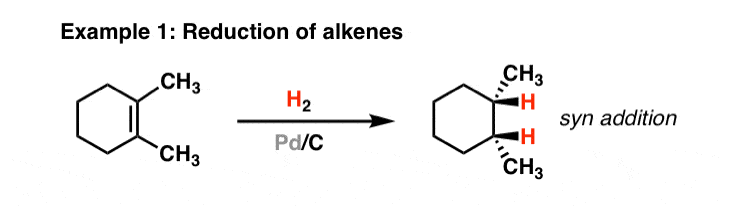
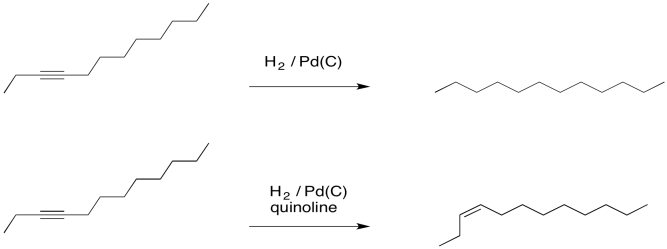
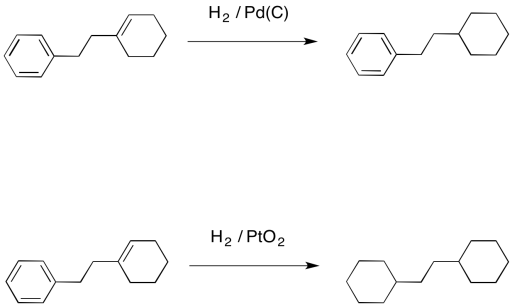
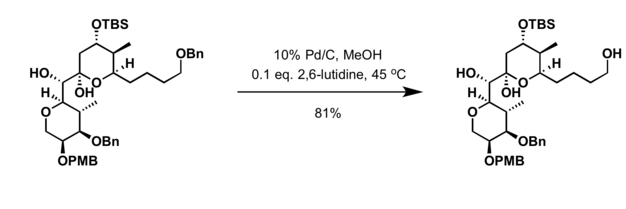
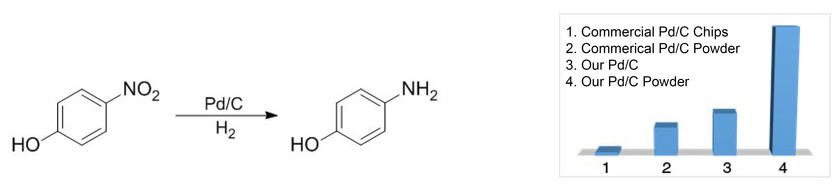
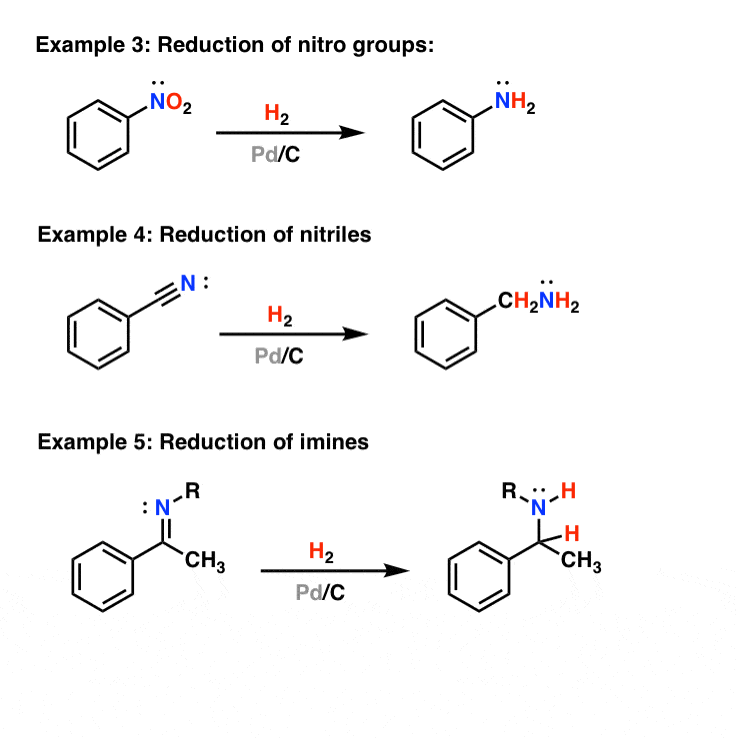
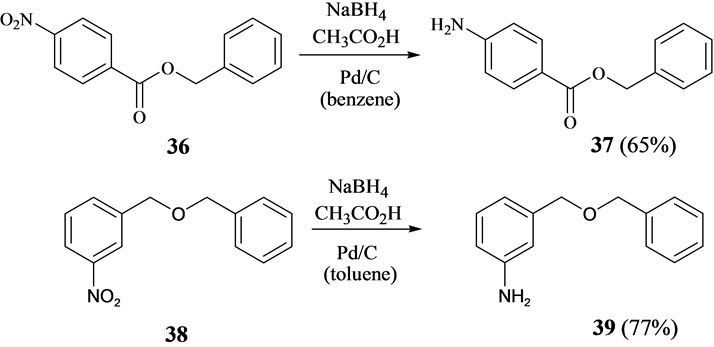Chemoselective control of hydrogenation among aromatic carbonyl and benzyl alcohol derivatives using Pd/C(en) catalyst - ScienceDirect
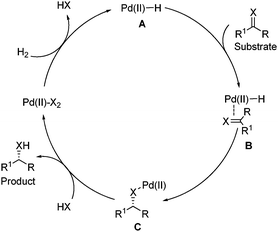
Homogeneous palladium -catalyzed asymmetric hydrogenation - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C2CS35333D

Development of a Palladium‐Catalyzed Process for the Synthesis of Z‐Alkenes by Sequential Sonogashira–Hydrogenation Reaction - Hancker - 2018 - European Journal of Organic Chemistry - Wiley Online Library
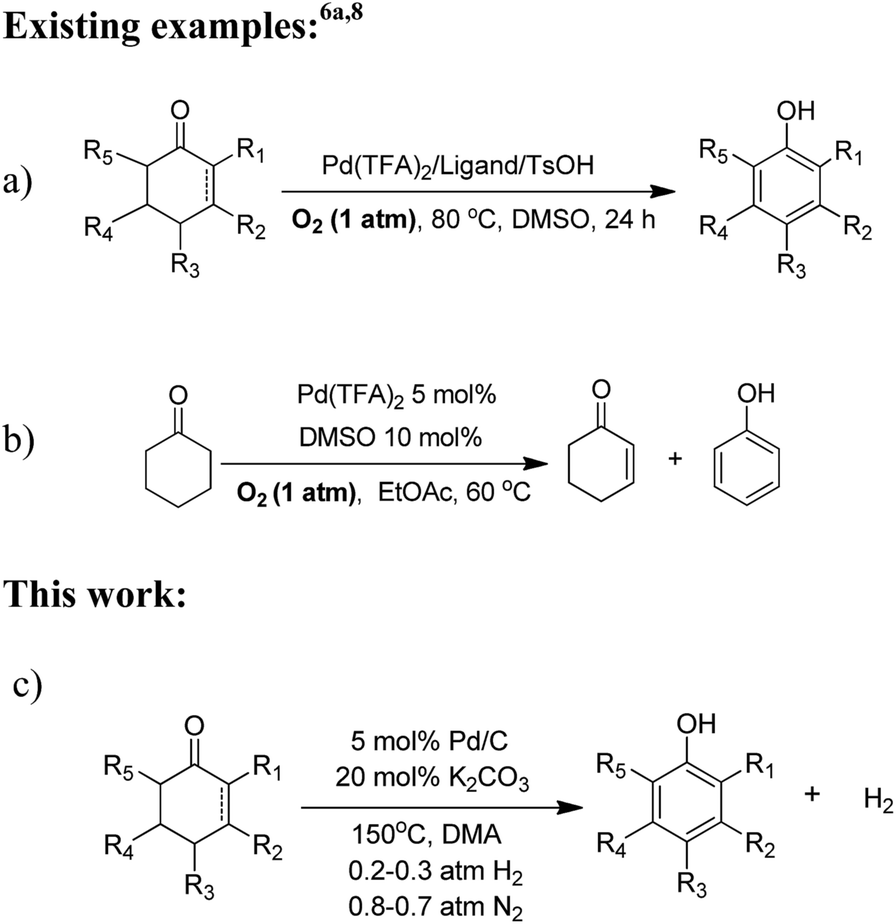
Reaction-activated palladium catalyst for dehydrogenation of substituted cyclohexanones to phenols and H 2 without oxidants and hydrogen acceptors - Chemical Science (RSC Publishing) DOI:10.1039/C5SC01044F

Hydrogenation of phenylacetylene and 3-ethynylanisole using palladium... | Download Scientific Diagram

PVA‐encapsulated Palladium Nanoparticles: Eco‐friendly and Highly Selective Catalyst for Hydrogenation of Nitrobenzene in Aqueous Medium - Wang - 2019 - Chemistry – An Asian Journal - Wiley Online Library
![Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/us/category/images/00074-img02.png)
Chemoselective Reduction catalysts | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation
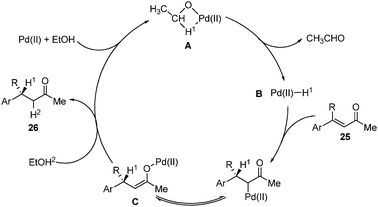
Homogeneous palladium -catalyzed asymmetric hydrogenation - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C2CS35333D
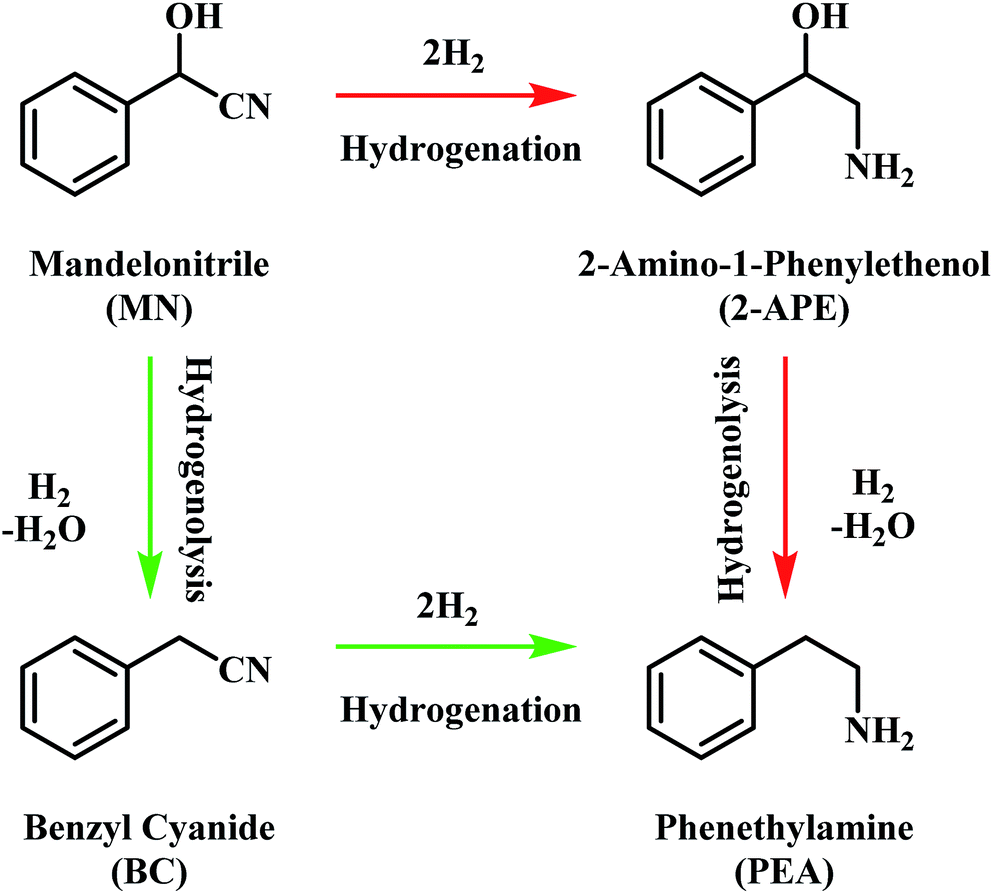
The hydrogenation of mandelonitrile over a Pd/C catalyst: towards a mechanistic understanding - RSC Advances (RSC Publishing)

PDF) Catalytic hydrogenation of acetylenic alcohols using palladium complex of fullerene C60 | Natalia Semagina - Academia.edu